MAX IV and ESS for life and medical science
MAX IV and ESS are two large research infrastructures that generates radiation in terms of photons (MAX IV) and neutrons (ESS) for imaging, spectroscopy and diffraction measurements at dedicated experimental stations. Get started with MAX IV and ESS!
European Spallation Source (ESS) is a European research infrastructure with contributions from 17 nations including Sweden. ESS will be the best neutron source ever constructed delivering an outstanding neutron intensity at the sample that combined with modern detectors will attract the best staff and research programs from the European neutron community to ESS. The ESS beamlines will be on average 20 times more intense than the current best neutron sources and some of the ESS beamlines more than 200 times better! meaning that measurements can then be done 20-200 times faster at ESS compared to any other neutron sources in the world. The ESS user program is estimated to start in 2027.
MAX IV is a Swedish synchrotron light source inaugurated in 2016 now with all planned beamlines available for research. Photons generated at MAX IV are exceptionally brilliant meaning that can generate many photons per seconds while photon angular divergence, beam cross-sectional area and photon frequency bandwidth are small. The high brilliance enables novel synchrotron radiation imaging and diffraction experiments when exposing biological samples at MAX IV.
Community and research highlights
- ESS related research highlights
- SwedNess - Swedish Neutron Education for Science and Society
- SNSS - Swedish Neutron Scattering Society
- LINXS - LINXS Institute of Neutron and X-ray Science
Coming events
More info on MAX IV and ESS
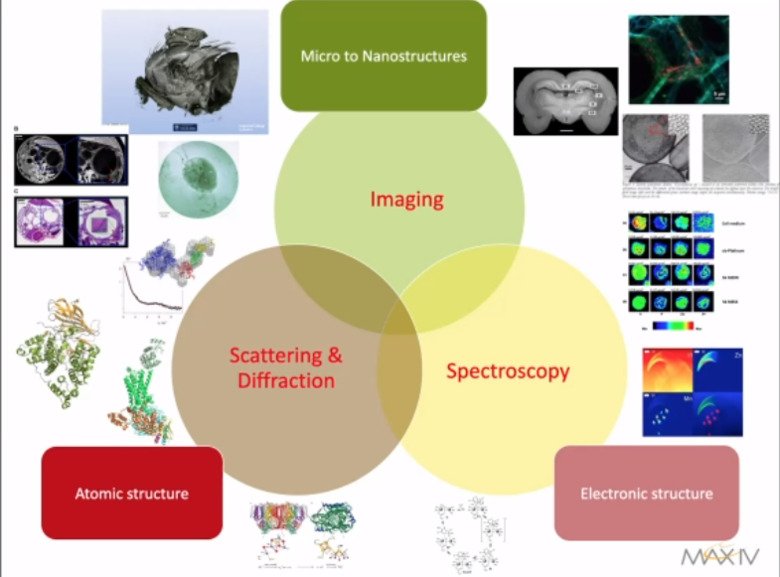
How to apply MAX IV and ESS in your research?
Experiments conducted at MAX IV and ESS are peer-reviewed and the experimental stations available represent a toolbox of novel opportunities that cannot be met elsewhere. Visiting scientists typically spend 24/7 of their time onsite engaging in spontaneous networking and crossbreeding of methods and ideas contributing to an ecosystem of activities surrounding large research infrastructures like MAX IV and ESS. New ESS and MAX IV users are advised to contact local staff to discuss project feasibility prior to write a specific MAX IV beamline, or ESS instrument, application for peer-review. Successful applicants can then use that beamline free of charge, however costs for visits and sending samples remain. Karolinska Institutet researchers can address complex research questions by exposing tissue and biological samples to these unique radiation sources and we can expect life and medical science taking significant steps forward.
ESS neutrons and MAX IV photons can be used for imaging, spectroscopy and scattering experiments. Please enjoy An overview of bioimaging beamlines at MAX IV shared by Karina Thånell. Below we will share links to events and information that might benefit researchers new to MAX IV and ESS. We need to familiarize ourselves with these tools to ask suitable research questions and prepare our samples accordingly. Below we present a selected set of MAX IV and ESS instruments related to life science and link to active KI researchers for guidance and collaboration. We also guide readers to local sample preparation facilities and other actors pushing for an increased use of large-scale research facilities for Swedish life-science.
Imaging at ESS and MAX IV
Imaging from the outside like a photography, or on the inside like 2D X-ray images or 3D representations made by tomography.
NanoMAX (MAX IV)
NanoMAX is a hard X-ray beamline (6-30 eV) with 30-300 nm beamsize for Scanning transmission microscopy (STXM) with absorption and phase contrast X-ray fluorescence microscopy (XRF) Coherent diffraction imaging techniques (CDI) Ptychography tomography1 in forward direction and Bragg geometry.
Ptychography tomography generates large dataset where the sample is rotated when illuminated by coherent X-rays and data from the detector transferred into a 3D image by computational methods. It is microscopy without probes and lenses where the extreme coherence of MAX IV will deliver excellent results. Initially ptychography has been used to study bone mineralization at MAX IV, and numerous life and medical science applications are expected.
Please enjoy X-ray phase contrast holographic nanotomography images from human diabetic peripheral nerves2 an note that NanoMAX staff is just about to publish first results from holography and holotomography performed at NanoMAX (in review)
Please listen to NanoMAX beamline online presentation from MAX IV user meeting 2020, review science case NanoMAX. NanoMAX also assembled a step-by-step guide to XRF imaging analysis.
1. First ptychographic X-ray computed tomography experiment on the NanoMAX beamline.
Kahnt M, Sala S, Johansson U, Björling A, Jiang Z, Kalbfleisch S, Lenrick F, Pikul JH, Thånell K
J Appl Crystallogr 2020 Dec;53(Pt 6):1444-1451
2. Three-dimensional architecture of human diabetic peripheral nerves revealed by X-ray phase contrast holographic nanotomography.
Dahlin LB, Rix KR, Dahl VA, Dahl AB, Jensen JN, Cloetens P, Pacureanu A, Mohseni S, Thomsen NOB, Bech M
Sci Rep 2020 05;10(1):7592
NanoMAX users at Karolinska Institutet
Karolinska Institutet Staff using NanoMAX today are:
Ptychography offers a range of opportunities for novel medical research so we expect to add more NanoMAX users from KI to the list above. First time NanoMAX users are strongly recommended to contact Simone Sala or other staff from NanoMAX to discuss feasibility, sample preparation and application procedures to become a NanoMAX beamline user.
SoftiMAX (MAX IV)
SoftiMAX is a soft X-ray beamline (275-2500 eV) for spectromicroscopy and coherent imaging suitable for a wide range of samples and research fields. Chemical contrast on a wide range of elements, including carbon, can be obtained by scanning the incoming photon energy over absorption edges. Samples need to be thin (100 nm - a few microns) and are usually supported on commercially available TEM grids or Si3N4 membranes, with a field of view of 1mm2.
SoftiMAX consists of two branches where the first branch is for Scanning Transmission X-ray Microscopy (STXM) and Ptychography using a focused 10-100 nm beam, while the second branch will host a more modularcoherent x-ray imaging (CXI) station using a larger beam size (20 x 20 micron) for scattering and full-field techniques like holography.
Under spring and autumn 2021 SoftiMAX will be commissioning the STXM branch with experts, and a first general user call is expected in autumn 2021. SoftiMAX is directly funded by the Swedish Research Council and a first way to learn about the beamline is to listen to online presentation by SoftiMAX staff from the 2020 MAX IV user meeting and feel free to contact the SoftiMAX staff to discuss your project.
ODIN (ESS)
Neutron imaging of the transitions required for life involving micro and nanostructures can be expected from ODIN. Please review the ODIN instrument proposal for details.
Spectroscopy at ESS and MAX IV
Light and matter are two representations of energy so how matter respond to illumination of various types of light is what spectroscopy is about. Neutrons have a spin-angular momentum (S=1/2) so a beam of neutrons may be polarized and interact with both nuclear and electronic magnetic moments in the sample. Please find an introduction to polarized neutron scattering.
T-REX (ESS)
T-REX is a bispectral chopper spectrophotometer intended for single crystals where both polarized and non-polarized neutrons can be measured. According to the T-REX proposal use of polarized photons give give the opportunity to separate coherent and incoherent signals on hydrogenated samples, avoiding H/D isotope exchange for this type of dynamic measurements in macromolecules.
CSPEC (ESS)
The Cold Chopper Spectrometer can be used to monitor the dynamic interplay between macromolecules and their hydrated shell1. Please review the CSPEC instrument proposal from 2013 and a recent review from 2020 on the CSPEC instrument available at another Spallation Neutron Source in Oak Ridge.
1. Dynamics of protein and its hydration water: neutron scattering studies on fully deuterated GFP.
Nickels JD, O'Neill H, Hong L, Tyagi M, Ehlers G, Weiss KL, Zhang Q, Yi Z, Mamontov E, Smith JC, Sokolov AP
Biophys J 2012 Oct;103(7):1566-75
Balder (MAX IV)
Balder is dedicated to the many variants of X-ray absorption spectroscopy (XAS) and X-ray emission spectroscopy (XES). Please listen to beamline staff presenting Balder from MAX IV user meeting 2020. Please review user information and get in contact with Balder staff for feasibility questions and preparations of biological samples.
Scattering at ESS and MAX IV
Scattering is the process by which light or other electromagnetic radiation is redirected in various directions when it encounters particles or obstacles in its path. Elastic scattering is when the incident light interacts with the material without changing its energy so that the scattered photons have the same energy as the incident photons. During inelastic scattering the scattered photons' energy differs from the incident photons, indicating a transfer of energy to the material.
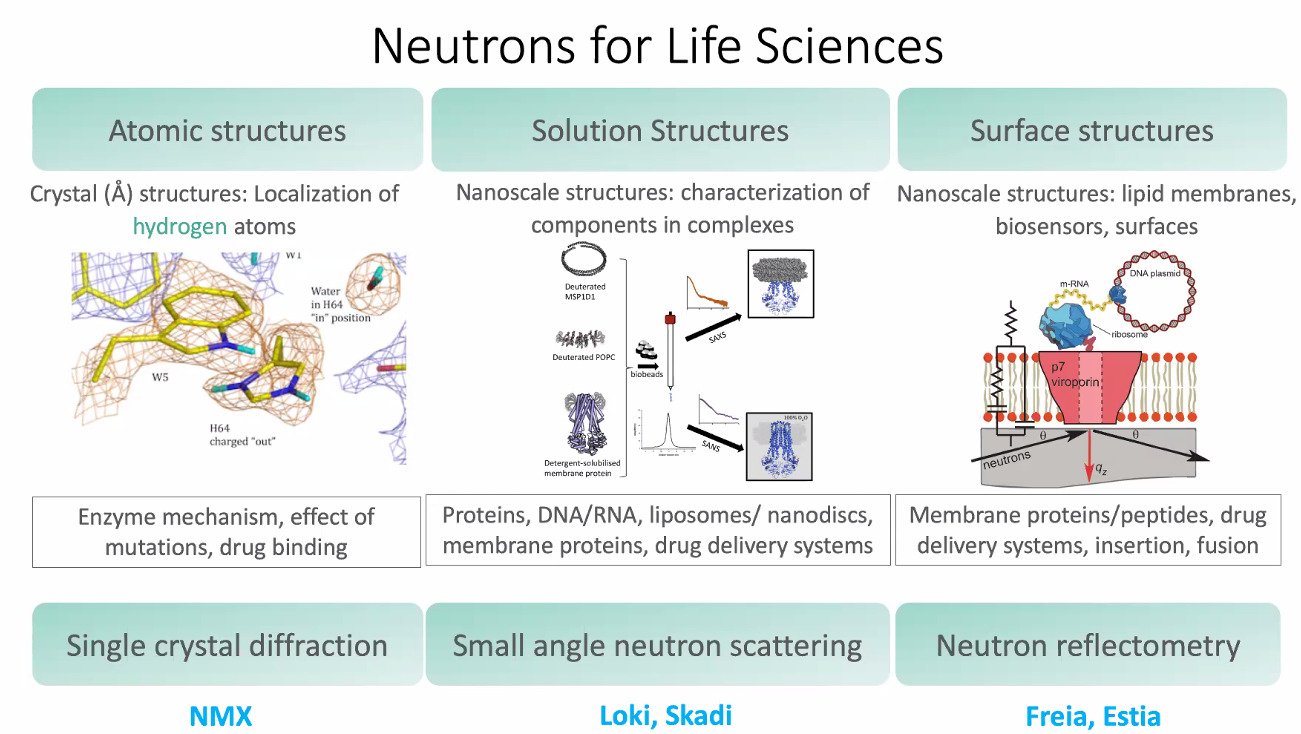
LOKI (ESS)
Small angle neutron scattering (SANS) was recently used by Astra Zeneca in determining the lipid composition of lipid nanoparticles1. Lipid nanoparticles are used frequently today as vehicles for mRNA Covid-19 vaccines from Pfizer/Biontech2. LOKI is constructed with support from ISIS and please review the LOKI proposal.
1. Successful reprogramming of cellular protein production through mRNA delivered by functionalized lipid nanoparticles.
Yanez Arteta M, Kjellman T, Bartesaghi S, Wallin S, Wu X, Kvist AJ, Dabkowska A, Székely N, Radulescu A, Bergenholtz J, Lindfors L
Proc Natl Acad Sci U S A 2018 04;115(15):E3351-E3360
2. mRNA-lipid nanoparticle COVID-19 vaccines: Structure and stability.
Schoenmaker L, Witzigmann D, Kulkarni JA, Verbeke R, Kersten G, Jiskoot W, Crommelin DJA
Int J Pharm 2021 May;601():120586
KI researchers using scattering at ESS and MAX IV
Hanna Barriga use both SAXS and SANS to study lipid nanoparticles with applications towards introducing RNA/DNA or drug molecules into cells.
Miracles (ESS)
Miracles is a time-of-flight backscattering instrument at ESS made by two teams in Bilbao and Madrid. Among other fields Miracles are also intended for life science such as molecular nanomagnets, molecular dynamics with implications to Alzheimer and Parkinson. Read more about how Miracles will be designed1 and a proposed upgrade to the initial plans
1. Conceptual design of the time-of-flight backscattering spectrometer, MIRACLES, at the European Spallation Source.
Tsapatsaris N, Lechner RE, Markó M, Bordallo HN
Rev Sci Instrum 2016 Aug;87(8):085118
CoSAXS (MAX IV)
CoSAXS (Coherent Small Angle X-ray Scattering) is a beamline designed for time-resolved studies of biological samples. At CoSAXS you may perform X-ray imaging, diffraction or spectroscopy experiments. Samples exposed to the X-ray beam before and after being heated by a second separate infrared laser beam enables temperature dependent phase transitions like macromolecule folding-unfolding or biological membrane dynamics to be measured.
The maximum sample-detector distance of 17 meters is ideal to perform small-angle diffraction measurements and CoSAXS will evolve to ultimately enable X-ray Photon Correlation Spectroscopy (XPCS) measurements - XPCS a new tool for life science.
Please listen to staff presenting CoSAXS from MAX IV user meeting 2020.
Diffraction at ESS and MAX IV
Particles or radiation being scattered by matter changes direction and sometimes wavelength, so called inelastic scattering. When the the matter interacting with incoming radiation is somehow structured in a regular pattern, such as the famous double-slit experiment, or like macromolecules arranged in a crystal, the scattered radiation will sums up in directions decided by the crystal lattice and we talk about diffraction instead of scattering.
Macromolecular X-ray crystallography (MX), an established method for 3D structure determination of macromolecules such as proteins, DNA and RNA. X-ray crystallography used high intensity photons being scattered from the electron cloud surrounding each atom of the macromolecules building up the crystal lattice, and while the hydrogen atom has a single electron, and the weakest electron affinity of all elements, X-ray crystallography are generally unable to detect electron density from hydrogen atoms. In contrast when using neutrons at ESS the incoming neutrons are scattered from the nuclei instead of the electron cloud as in X-ray crystallography. Neutron crystallography is therefore an excellent complement to X-ray crystallography and sometimes protein hydrogen for deuterium exchange is made, to optimize signal from deuterium instead of hydrogen.
NMX (ESS)
Frequently in preparing for neutron crystallography, well diffracting crystals are developed, and 3D structure determination performed using standard MX. Knowing the crystallization conditions, hydrogens are later partially or completely exchanged for deuterium. Complete H/D exchange require protein production in Heavy (D20) water, while partial exchange can be obtained by exposing purified protein or crystals to D20. DEMAX is an ESS sample preparation facility for biological and chemical deuteration where the final step of preparing suitable crystals for neutron diffraction experiments are performed.
Traditionally neutron diffraction has been made at room temperature however cryogenic cooling will be available at NMX. The high neutron flux of ESS will bring neutron crystallography a significant step forward.
For more NMX and DEMAX info, please contact Esko Oksanen science focus team coordinator for life science/structural biology or Zoe Fisher group leader for DEMAX
FREIA (ESS)
Neutron reflectometry can be used to study dynamic propeties of biological membranes, surfaces and layers. A pioneer study performed on a similar instrument at ILL were using samples prepared at DEMAX1.
1. Neutron reflectometry and NMR spectroscopy of full-length Bcl-2 protein reveal its membrane localization and conformation.
Mushtaq AU, Ådén J, Clifton LA, Wacklin-Knecht H, Campana M, Dingeldein APG, Persson C, Sparrman T, Gröbner G
Commun Biol 2021 Apr;4(1):507
BioMAX (MAX IV)
BioMAX is the first MX beamline at MAX IV presented on YouTube by beamline manager Ana Gonzales. Macromolecular crystals may also be used at FemtoMAX and upcoming MicroMAX.
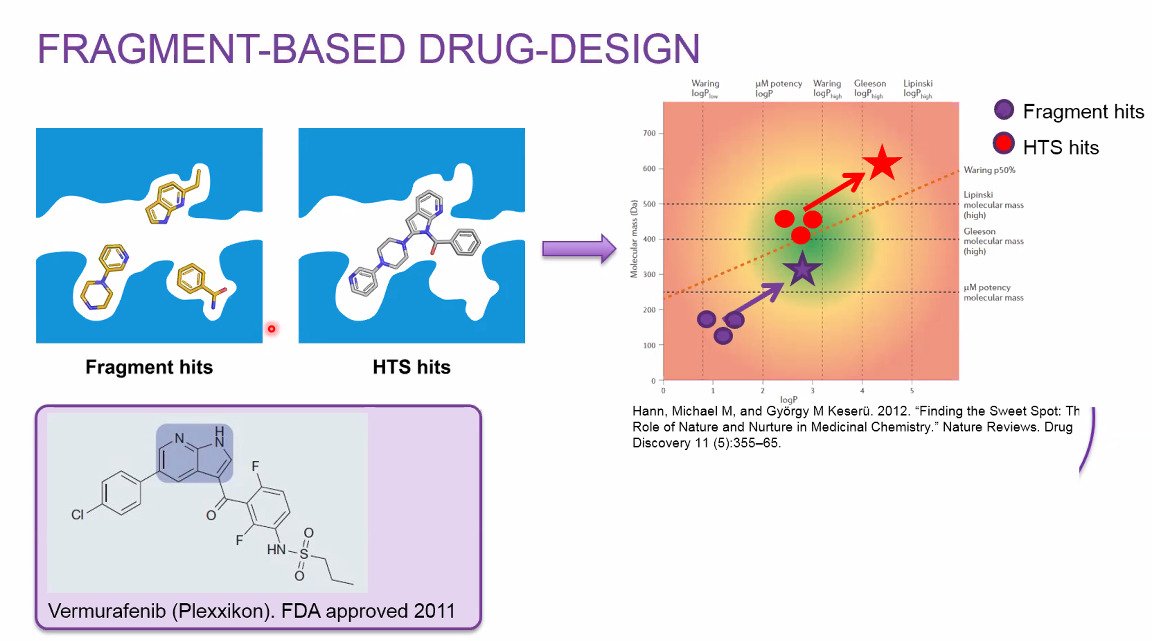
Structures of protein-ligand complexes are frequently determined by the pharmaceutical industry as a proof of target engagement of their small-molecule ligand drugs and FragMAX is a the BioMAX screening platform supported by Swedish pharma industry. FragMAX1,2 is not a beamline, instead it is an experimental setup dedicated towards 3D structure determination of very small ligand fragments and used to probe functional sites in proteins to ultimately develop novel and refined substances in early drug discovery.
1. FragMAX: the fragment-screening platform at the MAX IV Laboratory.
Lima GMA, Talibov VO, Jagudin E, Sele C, Nyblom M, Knecht W, Logan DT, Sjögren T, Mueller U
Acta Crystallogr D Struct Biol 2020 Aug;76(Pt 8):771-777
2. FragMAXapp: crystallographic fragment-screening data-analysis and project-management system. Lima, G. M. A., Jagudin, E., Talibov, V. O., Benz, L. S., Marullo, C., Barthel, T., Wollenhaupt, J., Weiss, M. S. & Mueller, U. Acta Cryst. 2021 D77.
KI researchers and core-facilities related to BioMAX
Macromolecular X-ray crystallography is used by many researchers at Karolinska Institutet and besides BioMAX staff you may want to contact one of the many local research groups using BioMAX for more info and perhaps collaborations:
Karolinska Institutet also hosts a dedicated core facility (Protein Science Facility) to assist all researchers going from gene or interest into 3D structure determination via cloning, protein expression, purification, crystallization, X-ray data collection, phasing, 3D model building and deposition in the Protein Data Bank. PSF assemble research output from local KI researchers in collectively applying for BioMAX beamtime shared and discussed at regular user meetings. Sample preparation, i.e. the early steps of molecular cloning, protein expression, purification and crystallization to obtain well-ordered crystals, is the key to a successful X-ray diffraction measurement at BioMAX or future MicroMAX.
FemtoMAX (MAX IV)
FemtoMAX is located at the linear accelerator of MAX IV and can generate short and very intense X-ray pulses that can be used for diffraction experiments. By combining X-ray pulses with a second source of laser light ultrafast processes in response to laser light can be studied. FemtoMAX has recently been used to monitor ultrafast movements in proteins1. Compared to X-ray Free Electron Lasers (XFEL) FemtoMAX has a much lower intensity so sample can be pulsed several times. Today the pulse frequency is 2 Hz and 10 Hz will soon be available.
Please enjoy FemtoMAX online presentation from MAX IV user meeting 2020.
1. High-resolution macromolecular crystallography at the FemtoMAX beamline with time-over-threshold photon detection.
Jensen M, Ahlberg Gagnér V, Cabello Sánchez J, Bengtsson ÅUJ, Ekström JC, Björg Úlfarsdóttir T, Garcia-Bonete MJ, Jurgilaitis A, Kroon D, Pham VT, Checcia S, Coudert-Alteirac H, Schewa S, Rössle M, Rodilla H, Stake J, Zhaunerchyk V, Larsson J, Katona G
J Synchrotron Radiat 2021 Jan;28(Pt 1):64-70
MicroMAX (MAX IV)
MicroMAX is intended for serial crystallography and time-resolved crystallography using microcrystals and BioMAX has been used to evolve sample delivery systems for MicroMAX3. MicroMAX has been open for user proposals since late 2023.
3. Current status and future opportunities for serial crystallography at MAX IV Laboratory.
Shilova A, Lebrette H, Aurelius O, Nan J, Welin M, Kovacic R, Ghosh S, Safari C, Friel RJ, Milas M, Matej Z, Högbom M, Brändén G, Kloos M, Shoeman RL, Doak B, Ursby T, Håkansson M, Logan DT, Mueller U
J Synchrotron Radiat 2020 Sep;27(Pt 5):1095-1102
Sample preparation facilities
Both ESS and MAX IV have local sample preparation facilities in near proximity to the measurement stations. The ESS Science Support Systems encompass seven different platforms for sample preparation for various types of neutron experiments.
Several sample preparation facilities are also available in Sweden for instance all scattering beamlines require crystals of macromolecules to be generated at:
- Protein Science Facility, MBB, Karolinska Institutet. Please contact PSF staff for questions
- Lund Protein Production Platform, Department of Biology, Lund University
and in the many research groups using X-ray crystallography spread across Swedish universities.
NMX Neutron crystallography diffraction at ESS require large deuterated crystals perhaps made at:
- Deuteration and macromolecular crystallization (DEMAX). For more info please contact DEMAX group leader Zoe Fisher.
where DEMAX is co-localized with the Lund Protein Production Platform. Please find a success story regarding DEMAX preparing samples for Neutron reflectometry measurements at ISIS.
MAX IV and ESS sample delivery projects
AdaptoCell is a key sample delivery development project where MAX IV staff will create a microfluidic flow-cell platform for novel ways of delivering samples to Balder, CoSAXS and MicroMAX beamlines
After beamtime data analysis
ESS will be supported by a dedicated data managment and software center (DMSC) in Copenhagen being the Danish in-kind contribution to ESS. Denmark also building a center for Quantification of Imaging Data from MAX IV (QIM). In similar manner MAX IV share a compute cluster between beamlines during data collection (online) and remote usage after beamtime (offline). MAX IV also invested in capacity for data management during the entire research life-cycle called DataStaMP.
Get involved with MAX IV and ESS
To optimize Swedish use of MAX IV and ESS, several initiatives have been taken in recent years some of which are already completed such as MAX4ESSFUN or in progress such as SWEDNESS graduate school for neutron research and Lund Institute of advanced Neutron and X-ray Science (LINXS) and the Office for MAX IV and ESS operated by Swedish Research Council and Vinnova.
LINXS
Lund Institute of advanced Neutron and X-ray Science (LINXS) is a source of knowledge for researchers that want to approach MAX IV and ESS. For instance, LINXS organizes
attempting to build a research community for neutron and X-ray science. LINXS divide activities at MAX IV and ESS into themes of research and todays themes are
Right now, LINXS has a call for new themes to be suggested by staff that belong to current LINXS partner organizations today being Lund University only.
Office for ESS and MAX IV
The Office for ESS and MAX IV aim to ensure that Swedish academy and industry uses MAX IV and ESS so that Sweden achieves best possible return of investments. The Office has created a first version of a National implementation plan (in Swedish) to be reviewed every year and use as their own guide document to achieve main goals and subsidiary goals stated in the plan.
InfraLife
InfraLife is a collaboration between SciLifeLab-ESS-MAX IV that aim to increase collaboration between these research infrastructures. Together one aim to address complex research questions of significant magnitude starting with a workshop on antibiotic resistance. Workshops and seminars to be arranged and joint hashtags on social media is available.
Past Events
- 15-17 Jan 2024: MAX IV User Meeting UM35
- 11-14 Jun 2023: Conference on Biology and Synchrotron Radiation
- 9 May 2023: 3D histology with x-ray micro-tomography
- 22 Aug - 2 Sep 2022: Integrative structural biology course
- 11-14 Jan 2022: Surface X-ray and Neutron Scattering
- Oct-Nov 2021: NNSP-SwedNess Neutron School online - registration deadline 10 Oct
- 25-27 Oct 2021: MAXIV User Meeting 2021 - registration deadline 18 Oct
- 21 Oct 2021 : Strategy workshop structural biology - registration
- 20 Oct 2021 : 12:45-14:00 MAX IV and ESS as engines for breakthrough science - registration page
- 20 Oct 2021 : 09:00-12:00 MAX IV synchrotron tomography workshop
- 14 Oct 2021 : ESS/MAX IV Online Summit - registration deadline Oct 12
- 17 June (14:00-15:00) FragMAX for crystal based fragment screening
- 9-11 June, Northern Lights on Food, Registration deadline 31 May
- 2 June (09:00-13-00) InfraLife Workshop: Antimicrobial resistance
- 1 June. Neutrons in Life Science and Biomaterials (14-17 June)
- 26 May 09:00 What can ESS and MAX IV do for Swedish life science?
